This user's guide is complementary to the original manual that describes the imaging procedure with MetaMorph. For multidimensional image acquisition, please refer to the original user's manual.
Before turning on the system, please make sure a proper sample adapter is already installed on the stage. If not, install it before powering on the system. If the microscope is already on, switch off the microscope (#6) before installing the sample adapter. Otherwise the motorized stage may be damaged.
- (Optional) Fluorescent X-cite light (#1) if fluorescent samples are imaged; otherwise leave if off
- Power strip on the shelf (#2)
- Computer (#3) and log in as 'lmcfuser'
- (Optional) Evolve EMCCD camera to the left of the microscope (#5) if you are going to use it; otherwise leave it off
- Microscope (#6)
- (Optional) CoolSNAP HQ2 camera to the right of the microscope (#7) if you are going to use it; otherwise leave if off
- Open ZEN 2.3 Pro
There are 4 stage inserts for holding different samples - a heated insert for a 35 mm glass-bottomed dish or a chambered glass coverslip, an insert for multi-well plates, a universal frame for holding a slide or a dish, and another universal frame with clamps for holding slides.
Nearly everything on the microscope is motorized so can be controlled either on the LCD touchscreen or through ZEN.
Objectives
|
Mag |
NA |
Oil? |
DIC? |
|
2.5x |
0.075 |
NO |
NO |
|
5x |
0.16 |
NO |
NO |
|
10x |
0.30 |
NO |
Ph1 |
|
20x |
0.8 |
NO |
DICII |
|
40x |
0.75 |
NO |
DICII |
|
100x |
1.4 |
Yes |
DICIII |
1. ZEN 2.3 Pro opens to 'Locate' page by default. Lower down the objective a bit before loading the sample; Position the sample right above the objective
2. Choose an objective lens from the drop-down list in the program (step 1), on the touch pad, or from the objective field on the right side of ZEN
3. Choose an illumination (step 2). The quick access buttons are available for BF, PH1, DICII, DICIII, DAPI, Cy5 (Far Red) , GFP, YFP, and Texas Red. The CFP and Cy7 (near-infrared) filter cubes are not installed, but are available upon request.
4. Coarse/fine focus knobs are on the microscope. X-Y stage movement is through the rotary controller. Focus on the sample; Change to a different objective if it is desired and fine tuning of the focus may be needed.
Basic Acquisition (Single Field):
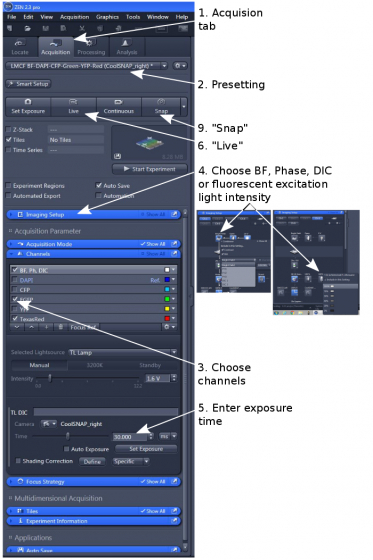
1. Load one of the 3 pre-settings dependent on your need (step 2). Attached to the left of the microscope is the higher sensitivity Evolve EMCCD and the higher resolution CoolSNAP HQ2 is on the right.
2. Open 'Channels' and tick all the channels you want to acquire images from (step 3)
3. Finding a good exposure for each channel; Don't use 'Auto Exposure' or 'Set Exposure' function
- For 'BF, Ph, DIC' channel:
(1) You need to use the objective-matching illumination (BF, phase contrast, DIC) under 'Imaging Setup' (step 4).
(2) Enter an exposure 'Time' and/or light 'Intensity' (step 5), start 'Live' (step 6); A live image will show.
(3) Adjust the exposure time and/or light intensity, so that the histogram of the image (showing under the live image, step 7) will extend to the right of the histogram box, but don't let the intensity of the brightest group of pixels exceed about 70% of maximum
(4) Optional - 'Shading Correction': move to an empty and clean area, still in live mode, click 'Define'. The system will acquire a shading correction reference image and activate the function.
- For fluorescent channels:
(1) Choose an exposure 'Time' (step 5) and/or an excitation light intensity (under 'Imaging Setup', step 4) and start 'Live' (step 6).
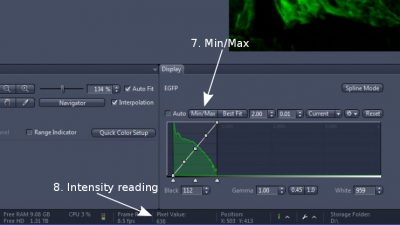
(2) Click 'Min/Max' or tick 'auto' in 'Display' section right below the image (step 7). This system adjusts the contrast of the image automatically for easy viewing of it.
(3) Hover the mouse pointer over your interested structure and read the intensity value near the bottom of the ZEN Window ('Pixel Value", step 8), and also read the intensity of a clean background. Adjust exposure time and/or excitation light intensity so that pixel value of the interested structure is 3 times or more over that of the background.
4. Click 'Snap' (step 9) to take images from all selected channels, and a merged image will be displayed. Individual channels can be separately displayed by clicking "Split"
5. Save images in ".czi" format
Tile-scanning:
Tile scanning of multiple continuous fields and stitching the tiles are used to generate a high-resolution image that covers an area larger than one field.
Method One: Quick tile image acquisition
This function is built in 'Snap' button. You can choose to 'Snap' single images (see above), or 2x2, 3x3, 4x4, 5x5 image tiles around the current position.
Method Two: Tile-scanning from a slide
Tick 'Tiles' to activate the function (step 1), determine a good exposure for each channel (see above) before proceeding to the next steps
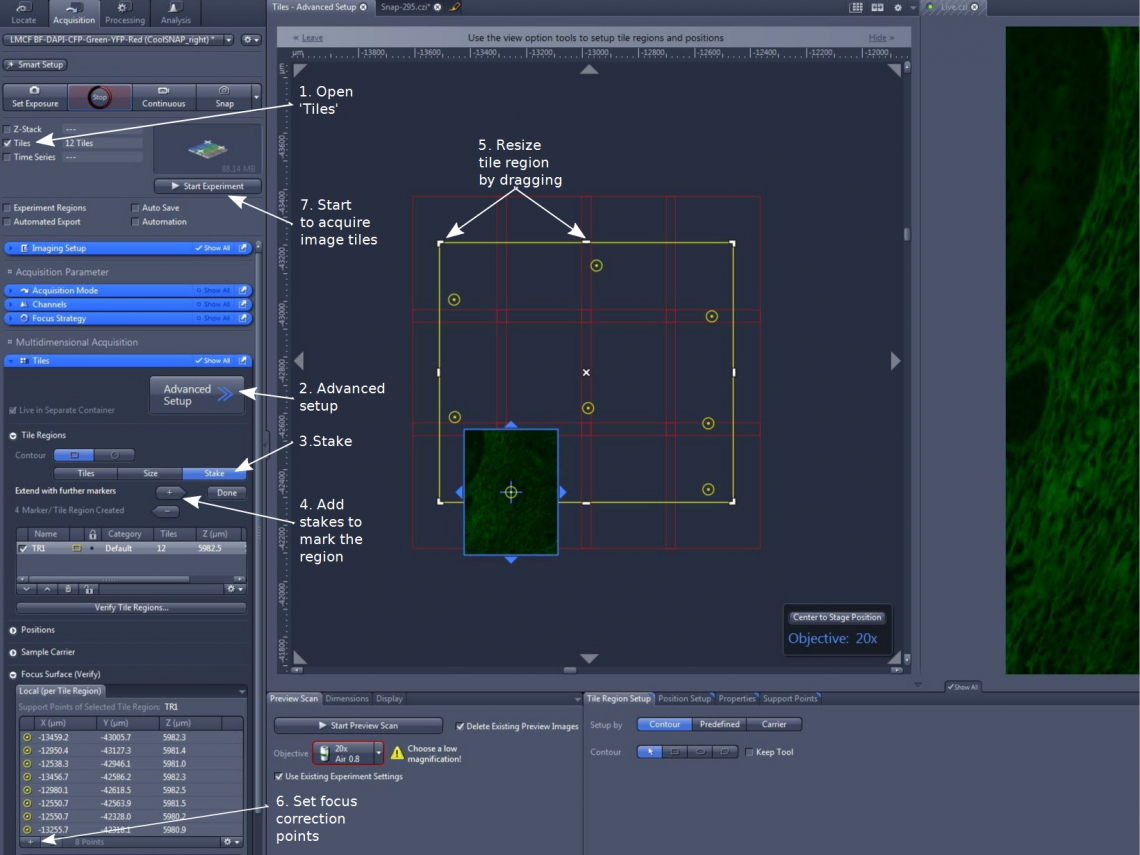
1. Under 'Tiles' section, click 'Advanced Setup' (step 2). The display window is divided into two parts for a virtual stage and a live image of the highlighted channel.
2. 'Tile Regions' set up: To mark the area to be imaged
(1). In 'Tile Regions', choose a 'Contour' and highlight 'Stake' (step 3)
(2). Move the sample stage and mark the stakes with the '+' sign (step 4). There need at least two diagonally positioned virtual 'stakes'. The system will draw a rectangle or a circle around them.
(3). The yellow line-enclosed area marks the desired area, while the red-line tiles will be the actual images to be taken. Check all areas outside the red tiles to make sure the desired imaging area is within the boundaries marked by red tiles. Otherwise, keep adding (the '+' sign) or drag the yellow line outwards (when cursor becomes a double head arrow, step 5). The area will be updated and expanded. To trim or reduce an area, drag the yellow line inwards
3. 'Focus Surface' setup: This is to set up focus-correction points that allows the user to manually set focuses at locations on the tissue section. The purpose is to 'flatten' out possibly uneven surface of the tissue section.
(1). Open 'Focus Surface (Verify)'
(2). Move the stage to a position, adjust focus, and add (using '+' sign) the x, y, and z information in memory (step 6)
(3). Repeat this step for more positions. Make sure the correction points cover the border area of the sample, and add more between them so that the points distribute evenly across the area. Through calculation, the software determines focuses for tiles not marked with focus-correction points.
4. Click 'Start Experiment' (step 7) to initiate tile acquisition
5. Stitch the tiles together (see below)
Method Three: Tile-scanning from wells in a multiwell plate or chambered coverglass
1. Tick both 'Tiles' and 'Auto Save' (step 1).
2. In 'Auto Save' (step 2), choose the folder that your data will be automatically saved in; Assign a base file name for the image tiles; Select ‘Close CZI Image After Acquisition’
3. In ‘Focus Strategy’ window (step 3), there are two options:
(1) 'Focus Surface and Global (Carrier Based)' is preferred for multiwell plates or chambered coverglass where the bottom of the vessel is assumed to be flat. In this mode, global support points are needed (see 6 below)
(2) 'Focus Surface and Local (per Region/Position)' allows setup of focus correction points in individual wells (see 10 below)
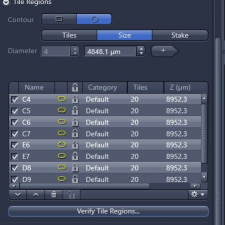
4. In ‘Tiles’ window, click to open ‘Tile Regions’, delete all previous tile regions if there are any in there (step 4);
In 'Options', make sure 'Split Scenes into Separate Files' is not selected
5. In 'Tiles' window, click to open 'Sample Carrier' and 'Select' a multiwell plate template (step 5);
6. The stage and the plate wells must be calibrated before acquiring images (step 6). ‘Calibrate’ wizard is to register the following:
(1) The upper-left corner of the plate: in wizard step 2/6, please find the upper left corner of the plate and click ‘Set Zero’. PLEASE DON’T CHOOSE ‘CALIBRATE’ because the stage starts moving immediately and may collide with the incubation chamber.
(2) The positions of the 'containers' (i.e. wells):
A. start ‘Live’ mode with a low-magnification lens (for example 5x lens) and bright field light, move to well 'A1'.
B. continue with calibration wizard, use 'Search Well Edge (7 Points)' for more accurate calibration
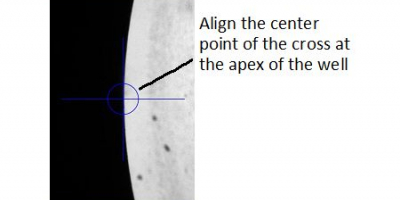
C. To 'Define Upper Well (A1)', make sure you align the center point of the cross to the apex of the edge of the well; Ticking 'Move Stage Automatically to each Required Position' allows the system to move to the next calibration point after 'Set Position' is clicked.
D. Continue until the calibration is 'finished'
7. Focus correction during image acquisition: in 'Tiles' window, assign global support points of the specific carrier (step 7) to compensate for inconsistent focuses across the plate:
Moving stage to the upper-left well (A1), clinking ‘+’ sign, and clicking the well on the multiwell template. Under 'Global Support Points', clicking to highlight the specific well , focusing on it, and clicking 'Set Current Z' to record the focus of the well. Repeat for other 3 corner wells.
It is strongly suggested to add more support points for better results. Even distance between the support points is preferred.
8. Click 'Advanced Setup' that splits the image window into a virtual stage and current live image window
9. Record the tile regions in wells:
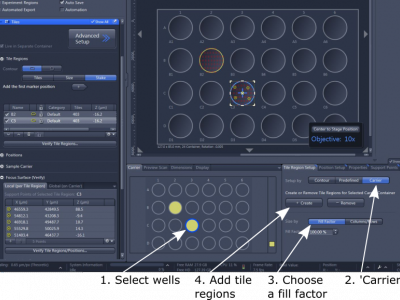
(1). In 'Carrier' under the image window and on the template, click the well that you wish to image
(2). In 'Tile Region Setup', choose 'Carrier'
(3) In 'Size By', choose 'Fill Factor 100% to cover the entire well; or other percentage to cover more or less than the entire well; or use 'Column x Row' to design the tile region
(4) '+ Create' to record the well in the 'Tile Regions'
(5) (Optional, but it is recommended), 'Start Experiment' to take images from this tile region (tiles in one well). That allows you to check the tile images and decide whether you are satisfied with the region setup.
(6) Repeat (1) to (4) to add more wells to the 'tile regions', or hold down ‘Shift’ key to select multiple wells across the rows and columns at once
10. (Alternative to 6 and better) Set up focus correction points ('Focus Surface (Verify)') for tile regain in individual well(s):
(1) Double-click a well on the template moves the well to imaging position, which also highlights the corresponding well in 'Tile Regions'; focus on the sample, then in 'Focus Surface (Verify)', add ('+' sign) the point (numbers in X, Y, and more importantly Z) in memory. It needs at least 4 focus points for the “Focus Surface” function to work.
(2) To add focus correction points to all selected wells at once, open 'Support Points' tab underneath the image window, select number of points in x and y, and 'Distribute' those support points to all wells that will be imaged. In 'Tile Regions' tab, start 'Verify Tile Regions' to set those support points
11. Select all or certain tile regions and ‘Start Experiment’
12. The final tile images from all wells are in a single file (scenes). They can be stitched all together (see below) and then split into individual images (scenes). (Alternatively, the tile images can be split into individual 'scenes' and tiles of each scene are stitched later (step 14))
13. To split the 'scenes' into separate files of individual wells:
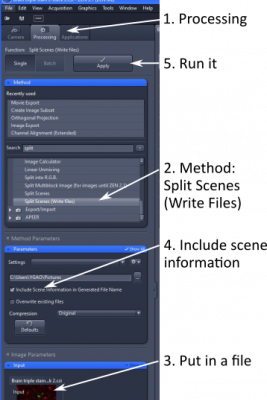
(1) Open 'Processing' tab
(2) Under 'Methods', find 'Utilities' and 'Split Scenes (Write Files)'
(3) Drop the 'scenes' file in 'input'
(4) In 'Parameter', check 'Include Scenes Information in Generated File Name' and choose a folder the resulting individual files go to
(5) 'Apply' to run the function
14. Stitch the tiles from individual tile regions one at a time or in batch
Stitching of Image Tiles:
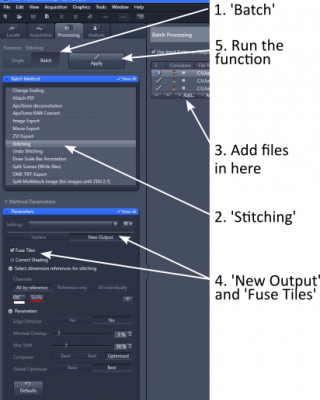
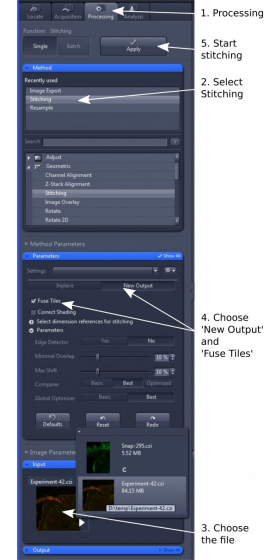
1. Open 'Processing' tab and select 'Stitching' (step 1 and 2)
2. In 'Input', choose the image tiles you wish to stitch (step 3)
3. In 'Parameters', choose 'New Output' and tick 'Fuse Tiles' (step 4). Other parameters are mostly default, but can be adjusted for best alignment and stitching.
4. Click 'Apply' to start the stitching process (step 5). The stitched image will be displayed as a separate image. Please save it in '.czi' format.
1. Contrast adjustment before image export:
For multichannel fluorescent images, the acquired images are shown as channel-merged. 'Split' the channels to display individual channels.
In 'Display', click the channel to be adjusted (step 1), drag the triangles under the histogram for white and black threshold to proper positions (step 2), so that the main structure is lightened up but not oversaturated and the background is dark and clean. Do the contrast adjustment in a sensitive and reasonable manner. Usually, the triangle for black point needs to be positioned right inside the main curve of the histogram. You can also double-click on a channel to have only the channel displayed, then hover the mouse cursor in the background area, read the intensity of the pixels (under 'Pixel Value'), type in the number as the background cut-off value (step 3).
Please keep in mind, adjustment of the contrast only affects the display of the image. It does not change the intrinsic intensity values of pixels in the image.
2. Image Export:
In 'Processing' tab, under 'Method', choose 'Image Export'; In 'Input', choose the image to be exported; In 'Parameters', use TIFF as file type (if a stitched file is too big, JPEG format or resizing the image is also acceptable). If you wish the exported images look as displayed, (for fluorescent images) tick 'Apply display Curve and Channel Color', 'Burn-in Graphics', 'Merged Channels Image', and 'Individual Channels Image' , then 'Apply' to let the image exported. Exporting fluorescent 'Raw Images' is optional.
Check (or re-check) the booking calendar online.
If somebody will use the microscope in one hour after you, clean the oil objective lens with lens tissue if you have used it, lower down the objective, close ZEN, leave the system on; If the person after you is not going to use the fluorescent light source (#1), turn it off; If the next user does not indicate whether he or she will use the fluorescent light, leave it on
If nobody comes to use the system in one hour, close the imaging program, turn the system off in the backward order of startup:
- CoolSNAP camera (#7) if you have used it
- Microscope (#6)
- Evolve EMCCD camera (#5) if you have used it
- Computer (#3)
- Power strip (#2)
- Fluorescent light source (#1)